42 balanced equation for baking soda and vinegar
How to Write the Net Ionic Equation for Baking Soda + Vinegar There are three main steps for writing the net ionic equation for Vinegar + Baking Soda (NaHCO3 + CH3COOH). First, we balance the molecular equation. Second, we write the states and... 11: Titration of Vinegar (Experiment) - Chemistry LibreTexts Sep 22, 2021 · Write the balanced equation for the neutralization reaction between aqueous sodium hydroxide and acetic acid. The Molarity of Acetic Acid in Vinegar Use your two best sets of results (with the palest pink equivalence points) along with the balanced equation to determine the molarity of acetic acid in vinegar.
Stoichiometry: Baking Soda and Vinegar Reactions - Stanford... baking soda to the soda bottle. This will allow you to measure the volume of air in the soda bottle. The volume of gas produced by the baking soda-vinegar reaction is equal to the volume of gas measured with the reaction minus the volume of gas measured without the reaction. 6. Repeat steps 3-10 with ½ tsp of baking soda.

Balanced equation for baking soda and vinegar
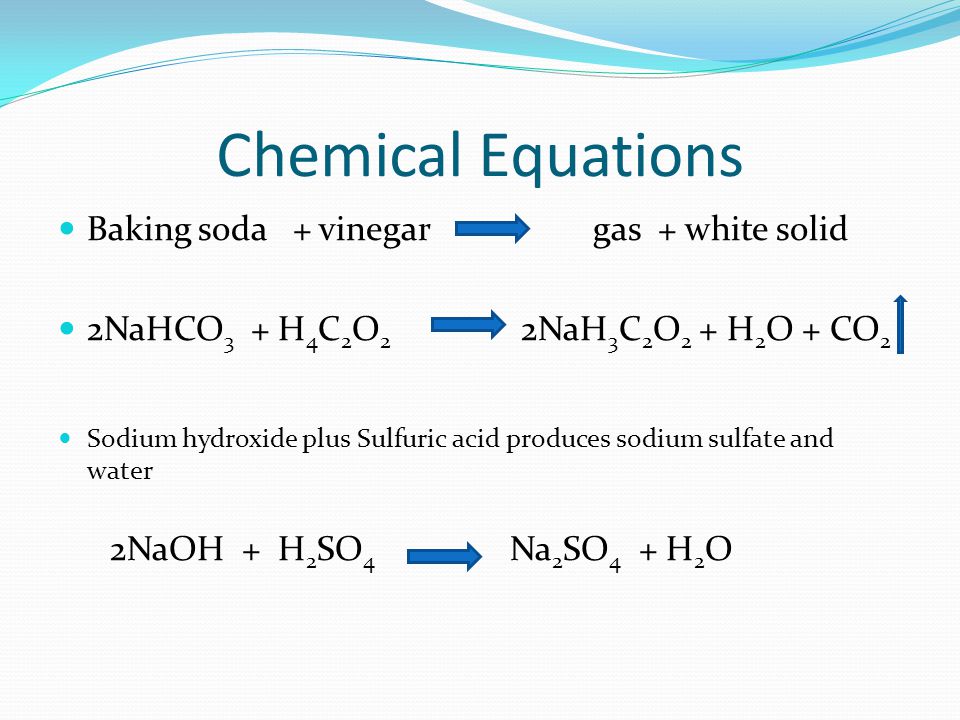
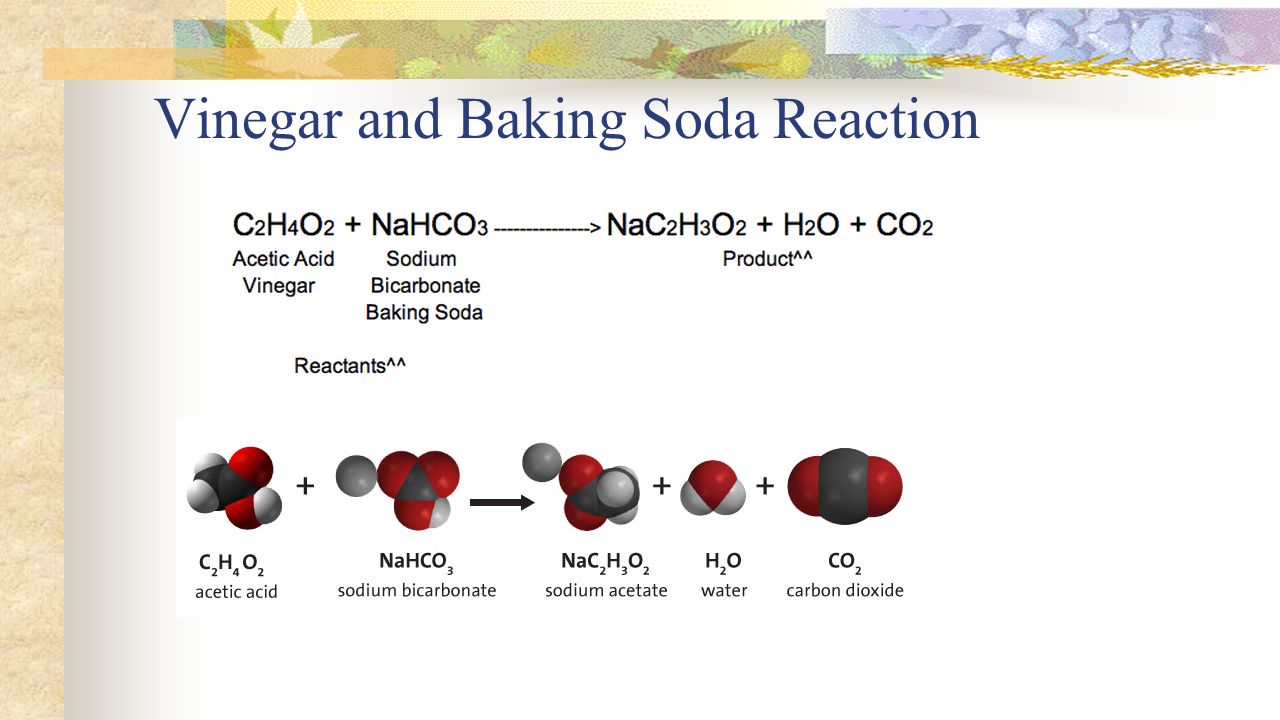
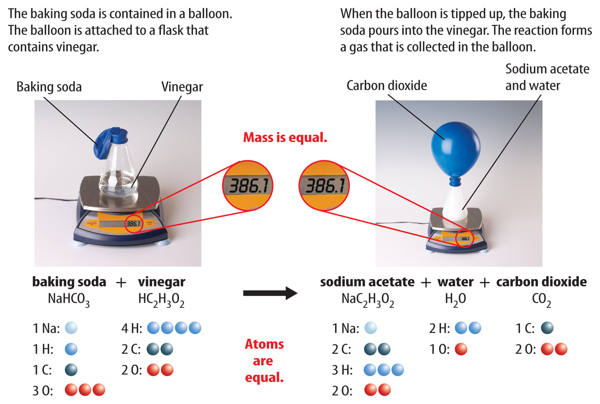
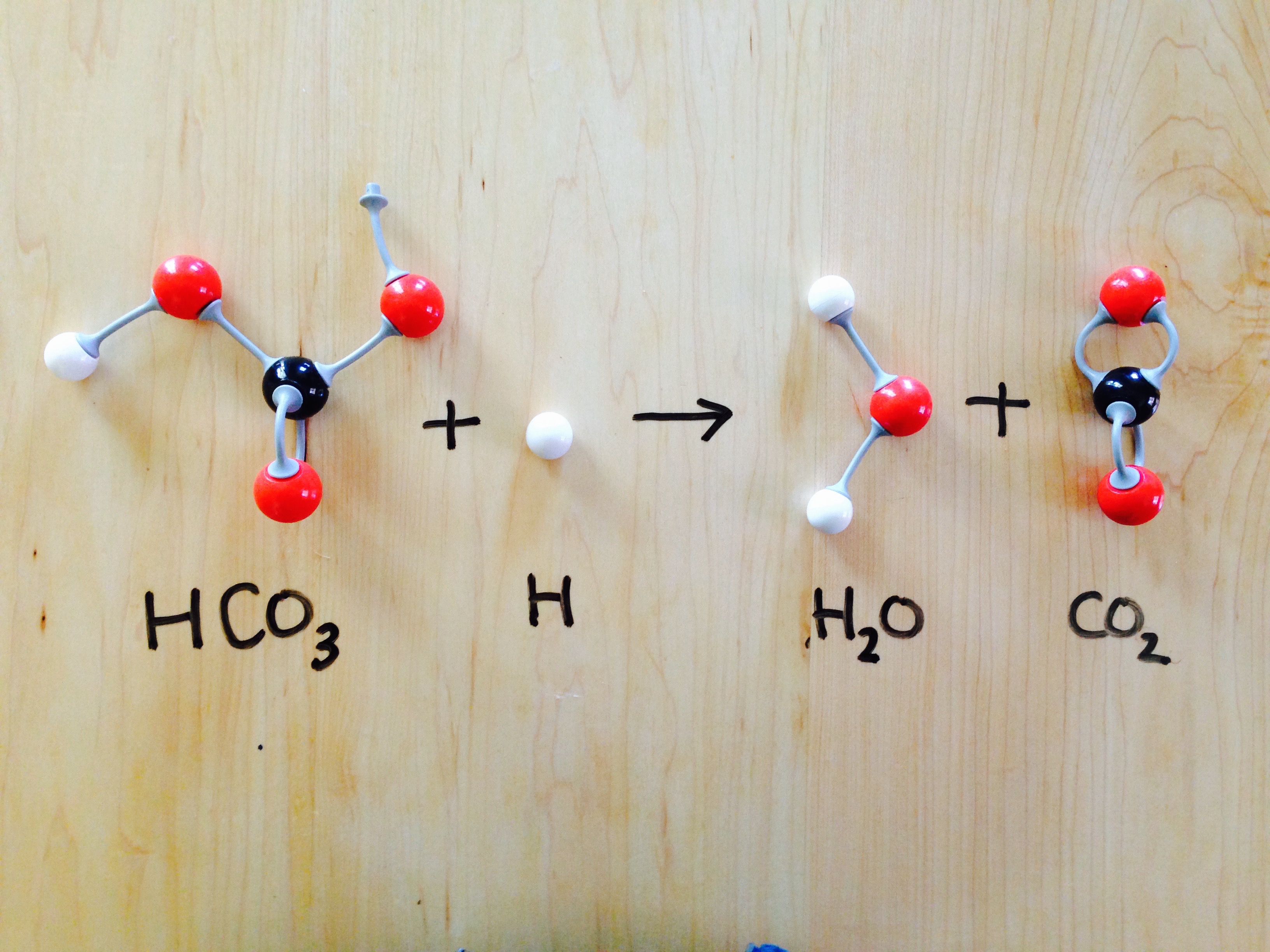
How to Balance: NaHCO3 + HC2H3O2 = NaC2H3O2 + CO2 + H2O|... Word equation: Sodium hydrogen carbonate + Acetic acid → Sodium acetate + Carbon dioxide + Water Type of Chemical Reaction: For this reaction we have a chemical reaction. Balancing Strategies: In this reaction we have NaHCO3 (baking soda) reacting with an aqueous solution of HC2H3O2 (vinegar) to form NaC2H3O2 + CO2 + H2O. Decomposition of Sodium Bicarbonate - Balanced Equation -... Dec 12, 2019 · The balanced equation for the reaction is: Na2CO3 (s) → Na2O (s) + CO2 (g) The decomposition of anhydrous sodium carbonate into sodium oxide and carbon dioxide occurs slowly at room temperature and proceeds to completion at 851 C (1124 K). Solved 11. Balance the chemical equation for the | Chegg.com Balance the chemical equation for the double-replacement reaction that occurred when baking soda was combined with vinegar. NaHCO3 (s) + + _HC2H302 (1) _NaC2H302 (aq) + _H2CO3 (aq) 12. One of the two products of the reaction of baking soda and vinegar is carbonic acid (H2CO3) which immediately forms water and a gas, Balance the equation showing the decomposition of carbonic acid _H2CO3 (aq) _____ H20 (1) +- _CO2 (9)
Balanced equation for baking soda and vinegar. Solved 11. Balance the chemical equation for the | Chegg.com Balance the chemical equation for the double-replacement reaction that occurred when baking soda was combined with vinegar. NaHCO3 (s) + + _HC2H302 (1) _NaC2H302 (aq) + _H2CO3 (aq) 12. One of the two products of the reaction of baking soda and vinegar is carbonic acid (H2CO3) which immediately forms water and a gas, Balance the equation showing the decomposition of carbonic acid _H2CO3 (aq) _____ H20 (1) +- _CO2 (9) Decomposition of Sodium Bicarbonate - Balanced Equation -... Dec 12, 2019 · The balanced equation for the reaction is: Na2CO3 (s) → Na2O (s) + CO2 (g) The decomposition of anhydrous sodium carbonate into sodium oxide and carbon dioxide occurs slowly at room temperature and proceeds to completion at 851 C (1124 K). How to Balance: NaHCO3 + HC2H3O2 = NaC2H3O2 + CO2 + H2O|... Word equation: Sodium hydrogen carbonate + Acetic acid → Sodium acetate + Carbon dioxide + Water Type of Chemical Reaction: For this reaction we have a chemical reaction. Balancing Strategies: In this reaction we have NaHCO3 (baking soda) reacting with an aqueous solution of HC2H3O2 (vinegar) to form NaC2H3O2 + CO2 + H2O.





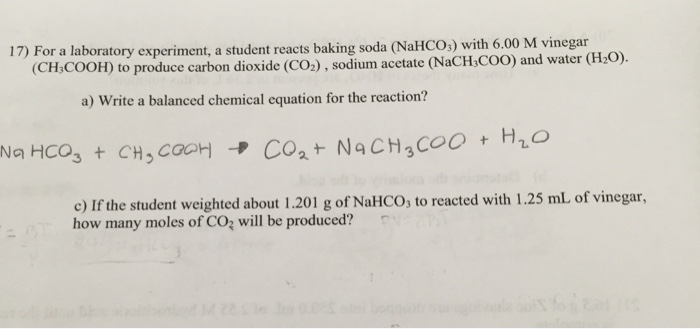







:max_bytes(150000):strip_icc()/acetic-acid-molecule-536230016-57b4825d5f9b58b5c2bdcfe1.jpg)








:max_bytes(150000):strip_icc()/aceticacid-56a129995f9b58b7d0bca2c4.jpg)
![ANSWERED] Acetic acid (CH3COOH) is the acid in vine ...](https://media.kunduz.com/media/sug-question/raw/60677547-1657054377.0366533.jpeg)







0 Response to "42 balanced equation for baking soda and vinegar"
Post a Comment